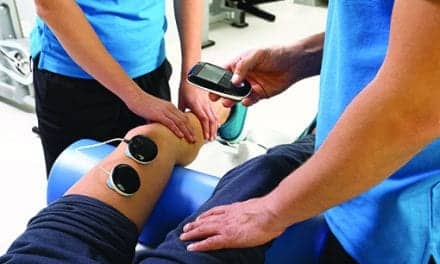
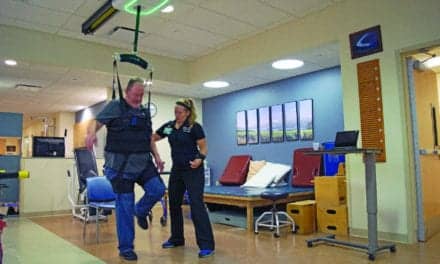
Overground robotic exoskeletons are worn on the outside of the user’s body and expand the therapist’s options for exercise and gait retraining. Pictured here, Clare Hartigan, PT, MPT, works with patient Patti Wise at Shepherd Center on improving gait and stability.
By Clare Hartigan, PT, MPT
Advancements in technology provide a wide range of systems to assist patients to walk. Although upright mobility is often the goal, secondary health benefits can be realized in combination with locomotor training. Health benefits from not being sedentary are indisputable. Technologies to promote walking in persons with neurological diagnoses have specifically shown corresponding positive changes in cardiovascular status, improved bowel/bladder function, decreased rates of infection, reduction in pain, lowered levels of muscle spasticity, increased bone density, reduction in blood lipid levels, decreased fat mass, increased muscle mass, overall weight loss, and enhanced psychological well-being.1-5
Of great debate within the rehabilitation profession is which approach provides the optimal environment to promote restorative walking function. Among the many systems to choose from, there are two main divisions: treadmill-based or overground. Systems to be reviewed in this article will include a sample of treadmill and overground powered devices proven to be safe, offer task-specific repetitive practice, provide opportunities for use-dependent plasticity, attempt to duplicate “normal walking” patterns, and encourage principles of motor learning. Neuro-prosthetics will not be addressed.
[sidebar float=”right” width=”250″]Product Resources
The following companies offer robotic technologies for rehabilitation:
Ekso Bionics
www.eksobionics.com
Gorbel Inc-Medical Division/SafeGait
www.safegait.com
Hocoma
www.hocoma.com
Mobility Research
www.Litegait.com
Parker Hannifin
www.indego.com
ReWalk Robotics
www.rewalk.com
Rex Bionics
www.rexbionics.com[/sidebar]
Feet On the Ground
Treadmill-based systems which allow for either manual leg assist or robotic leg assist in combination with body weight support harnesses are not new to rehabilitation. Systems like the Lokomat by Hocoma Inc, Norwell, Mass; the Therastride by Innoventor Inc, Earth City, Mo; the AutoAmbulator by Encompass Health, Birmingham, Ala; and others have been successful in providing gait rehabilitation to neurological patients with significant paralysis. Treadmill training with a body weight support harness certainly allows repetitive stepping without the need for compensatory braces (short or long leg braces), which may limit normal ranges of joint motion, varied amounts of weight to be placed on their legs rather than putting weight through the arms to manage a stability aid, and to have mass practice at varying speeds and muscular control.
Non-treadmill-based overhead harness body weight support systems are another group of popular technologies used for locomotor training. Such overground devices commonly found in rehabilitation centers include the ZeroG by Aretech, Ashburn, Va; the Vector by Bioness, Valencia, Calif; the Andago by Hocoma, Norwell, Mass; the LiteGait by Mobility Research Inc, Tempe, Ariz; the SafeGait 360 by Gorbel Medical, Victor, NY; and others. These systems were developed for patients to increase exploration of their environment, to provide additional challenges, to encourage task-specific exercise/gait training, and to be able to walk over a variety of “real life” surfaces.
Examining Exoskeletons
Overground Robotic Exoskeletons (ORE) are relatively new to neuro-rehabilitation and combine both robotic technology and the ability to walk overground. This type of technology is worn on the outside of the body and has been developed to provide options for exercise, gait retraining, and personal use. ORE in the United States are labeled as either Class 1 or Class 2 medical devices. Class 1 ORE devices are approved for exercise only and include the Rex by Rex Bionics, Boston; and the HAL by Cyberdyne. Class 2 ORE devices are approved for both exercise and gait rehabilitation and include the EksoGT by Ekso Bionics, the Indego by Parker Hannifin, and the ReWalk by ReWalk Robotics.
Given that many clinicians may not be familiar with class 2 OREs, a closer look at EksoGT, Indego, and ReWalk will provide them valuable information. The first question often asked regarding OREs is, what type of patients are appropriate to use such advanced technology? Each ORE device has slightly different requirements, but in general, patients should present with neurological injury or diagnosis which limits the ability to walk, weight below 221 lbs, approximate height of 5 feet 1 inch to 6 feet 3 inches, intact skin, passive range of motion to be within functional limits, healthy bone density, stable cardiovascular health, sufficient upper extremity strength to manage stability aids, and the ability to follow directions and to communicate basic needs.
With the ability to adjust levels of robotic assist (right or left sides) according to each patient’s unique motor scores, to use a variety of stability aids for functional progression (platform walkers, walkers, forearm crutches, or canes) and to incorporate normal walking patterns (four-point or two-point step timing), Ekso GT and Indego are designed to provide locomotor training across the continuum of care. Both systems are FDA approved for persons with spinal cord injury and stroke and may be incorporated as early as inpatient and all the way through to outpatient fitness programs. Skilled therapy billing codes which already exist and would be appropriate for ORE treatments include gait training, neuromuscular re-education, therapeutic exercise, and activities of daily living training.
Finding the Right Technology
Please consider the following three steps when trying to determine the most appropriate technology: 1) Form a team of therapists, physicians, and administrators to thoroughly review each system to determine which system fits both short-term and long-term goals of your program(s); 2) Establish “key users” of the technology who will provide efficient and high-quality interventions; and 3) Monitor outcomes for functional restoration, patient satisfaction, and program improvement.
Use of ORE technology outside of a rehabilitation setting is limited to patients with thoracic-level spinal cord injury (SCI) and to only two FDA-approved devices: Indego (T3 SCI or lower) or ReWalk (T7 SCI or lower). Conventional orthotics for people with thoracic-level SCI include a variety of reciprocal gait orthoses, long-leg braces, or short-leg braces, which, depending on their amount of lower-extremity muscle preservation, must also be used in combination with a stability aid. Given the amount of effort it takes for persons with thoracic-level SCI to walk with braces, they are often limited in walking time, distance, and environments due to fatigue or pain. ORE devices are not intended to replace a wheelchair as the primary means of mobility. However, given their ease of use with an appropriate stability aid, approximation to more normal joint motion during swing, and stance phases of gait, ability to walk over even/uneven surfaces, attainable walking speeds, and the ability to manage inside and outside terrains, OREs provide a functional and safe alternative to brace walking.6-8
Robotics in the Home
If a person wishes to obtain an ORE for home/community use, the FDA requires that both the user and a support person successfully complete a company-certified personal use training program and that the trained support person must be with the user at all times for safety. OREs are expensive to purchase. Therefore, it is highly recommended that one should trial both systems prior to making a decision about which device may best meet their expectations for intended use. A person would never purchase a car without test driving it first. The same should apply to OREs. A question often asked is, are personal use OREs covered by insurance? The Veterans Administration is the only organization which has publicly announced it will cover the cost of a device (Indego or ReWalk) and all necessary training hours for qualified veterans. Some private insurance companies have been willing to apply physical therapy benefits to cover some of the training hours, but not the cost of the actual device. PTP
Clare Hartigan, PT, MPT, is the program manager for Lower Extremity Robotics at The Virginia C. Crawford Research Institute, Shepherd Center, Atlanta. Hartigan received a Bachelor of Science in Biology, Cum Laude, from Bucknell University in 1986 and a Master in Physical Therapy, Highest Honor, from Emory University in 1989. Since 1991, she has been employed at Shepherd Center. Hartigan is a member of the American Physical Therapy Association and the Neurologic Section. For more information, contact [email protected].
References
1. Semerjian T, Montague S, Dominguez J, Davidian A, De Leon R. Enhancement of quality of life and body satisfaction through the use of adapted exercise devices for individuals with spinal cord injuries. Top Spinal Cord Inj Rehabil. 2005;11(2):95–108.
2. Hicks AL, Ginis KA. Treadmill training after spinal cord injury: it’s not just about the walking. J Rehabil Res Dev. 2008;45(2):241–248.
3. Karelis AD, Carvalho LP, Castillo MJ, Gagnon DH, Aubertin-Leheudre M. Effect on body composition and bone mineral density of walking with a robotic exoskeleton in adults with chronic spinal cord injury. J Rehabil Med. 2017;49(1):84–87.
4. Miller LE, Zimmermann AK, Herbert WG. Clinical effectiveness and safety of powered exoskeleton-assisted walking in patients with spinal cord injury: systematic review with meta-analysis. Med Devices (Auckl). 2016;9:455–466.
5. Stampacchia G, Rustici A, Bigazzi S, Gerini A, Tombini T, Mazzoleni S. Walking with a powered robotic exoskeleton: Subjective experience, spasticity and pain in spinal cord injured persons. NeuroRehabilitation. 2016;39(2):277–283.
6. Asselin PK, Avedissian M, Knezevic S, Kornfeld S, Spungen AM. Training persons with spinal cord injury to ambulate using a powered exoskeleton. J Vis Exp. 2016;112.
7. Hartigan C, Kandilakis C, Dalley S, et al. Mobility outcomes following five training sessions with a powered exoskeleton. Top Spinal Cord Inj Rehabil. 2015;21(2):93–99.
8. Louie DR, Eng JJ, Lam T. Gait speed using powered robotic exoskeletons after spinal cord injury: a systematic review and correlational study. J Neuroeng Rehabil. 2015.