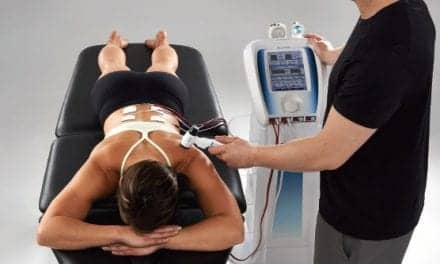
ManaMed announces the US Food and Drug Administration (FDA) approval for ManaSport, a new ultrasound therapy Class II device that provides pain relief and treatment for soft-tissue injuries and is popular among athletes and sports professionals. Safe and easy to use, the new device is set to offer life-changing support to those suffering from ongoing issues. FDA approval has now cleared the product for a range of indications for use, including:
- Chronic pain relief.
- Muscle spasms and joint contractures.
- Relief from pain and joint contractures associated with adhesive capsulitis, bursitis with slight calcification, myositis, soft tissue injuries, and shortened tendons from past injuries and scar tissue.
- Relief from pain and muscle spasms resulting from capsular tightness and capsular scarring.
- Increasing localized blood flow.
- Increasing the range of motion of contracted joints with heat and stretch techniques.
Portable and rechargeable, the device can now be prescribed by medical professionals and is intended for supervised use. It provides non-invasive pulsed ultrasound therapy at localized sites through ultrasound gel, with no discomfort to the patient. A complete therapy session takes just 20 minutes, and the product comes complete with everything you need to carry out therapy sessions, including ultrasound gel, a strap to hold the applicator in place, and a battery charger.
This compact medical device has an intuitive display with simple instructions for use. A built-in calendar helps users stay on top of treatments, while special safety features can lock the device to avoid unintended operation. Physicians and physiotherapists can manually move the applicator throughout the affected area, or use the included strap for a static, hands-free treatment.
Trevor Theriot, President, and CEO commented, “ManaMed will continue to design, develop, and innovate the entire Med Tech space. ManaSport demonstrates our commitment to invest in the future and expand our access to the entire continuum of care.”
[Source(s): ManaMed Inc, PR Newswire]